Introduction to Moles
👁 2 مشاهدات

النص الكامل للفيديو
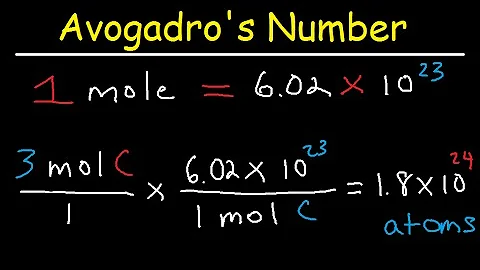 17:59
17:59
Avogadros Number The Mole Grams Atoms Molar Mass Calculations Introduction
The Organic Chemistry Tutor
3.9M مشاهدة · 9 yr ago
 7:15
7:15
Mole Conversions Made Easy How to Convert Between Grams and Moles
ketzbook
1M مشاهدة · 9 yr ago
 12:04
12:04
Mole Problems
thecollegegateway
3.5K مشاهدة · 13 yr ago
 19:15
19:15
Intro to Mole Problems
thecollegegateway
306 مشاهدة · 7 yr ago
 10:12
10:12
Very Common Mole Questions
Tyler DeWitt
456.4K مشاهدة · 12 yr ago
 9:22
9:22
How to answer any MOLES Chemistry question
Science Shorts
15.2K مشاهدة · 3 mo ago
 7:09
7:09
Step by Step Stoichiometry Practice Problems How to Pass Chemistry
Melissa Maribel
1.7M مشاهدة · 8 yr ago
 1:19:46
1:19:46
The ONLY Lesson ON mole Concept Full Lesson
The Dms Online School
90.5K مشاهدة · 2 yr ago
 5:29
5:29
How to calculate the number of moles Chemistry
Najam Academy
406.4K مشاهدة · 4 yr ago
 32:07
32:07
CHEMISTRY TEST REVIEW OVER MOLES UNIT
Mrs. Clarke's Chemistry Class
2.5K مشاهدة · 4 yr ago
 1:00
1:00
How to do any mole calculation for GCSE Chemistry summarised in 95 seconds
Flash Revision Lab
28.4K مشاهدة · 2 yr ago
 16:41
16:41
How to Calculate the Mole 5 Practice Problems 1 step and 2 step problems
Straight Science
5.6K مشاهدة · 5 yr ago
 8:46
8:46
Molarity Made Easy How to Calculate Molarity and Make Solutions
ketzbook
1.3M مشاهدة · 9 yr ago
 4:45
4:45
A Simple way to work out moles Chemistry past paper question answer
The Animated Chem Tutor
4.1K مشاهدة · 5 yr ago
 3:00
3:00
Mole Concept in 3 mins Simple
Amplify Learning - with Alok
82.5K مشاهدة · 7 mo ago
 25:16
25:16
Stoichiometry Basic Introduction Mole to Mole Grams to Grams Mole Ratio Practice Problems
The Organic Chemistry Tutor
5.4M مشاهدة · 8 yr ago
 12:11
12:11
Stoichiometry Mole to Mole Conversions Molar Ratio Practice Problems
The Organic Chemistry Tutor
1.1M مشاهدة · 8 yr ago
 6:06
6:06
The Mole Avogadros Number and Stoichiometry
Professor Dave Explains
835.5K مشاهدة · 10 yr ago
 0:11
0:11
Mole Calculations Explained
Matt Green
83K مشاهدة · 3 yr ago







